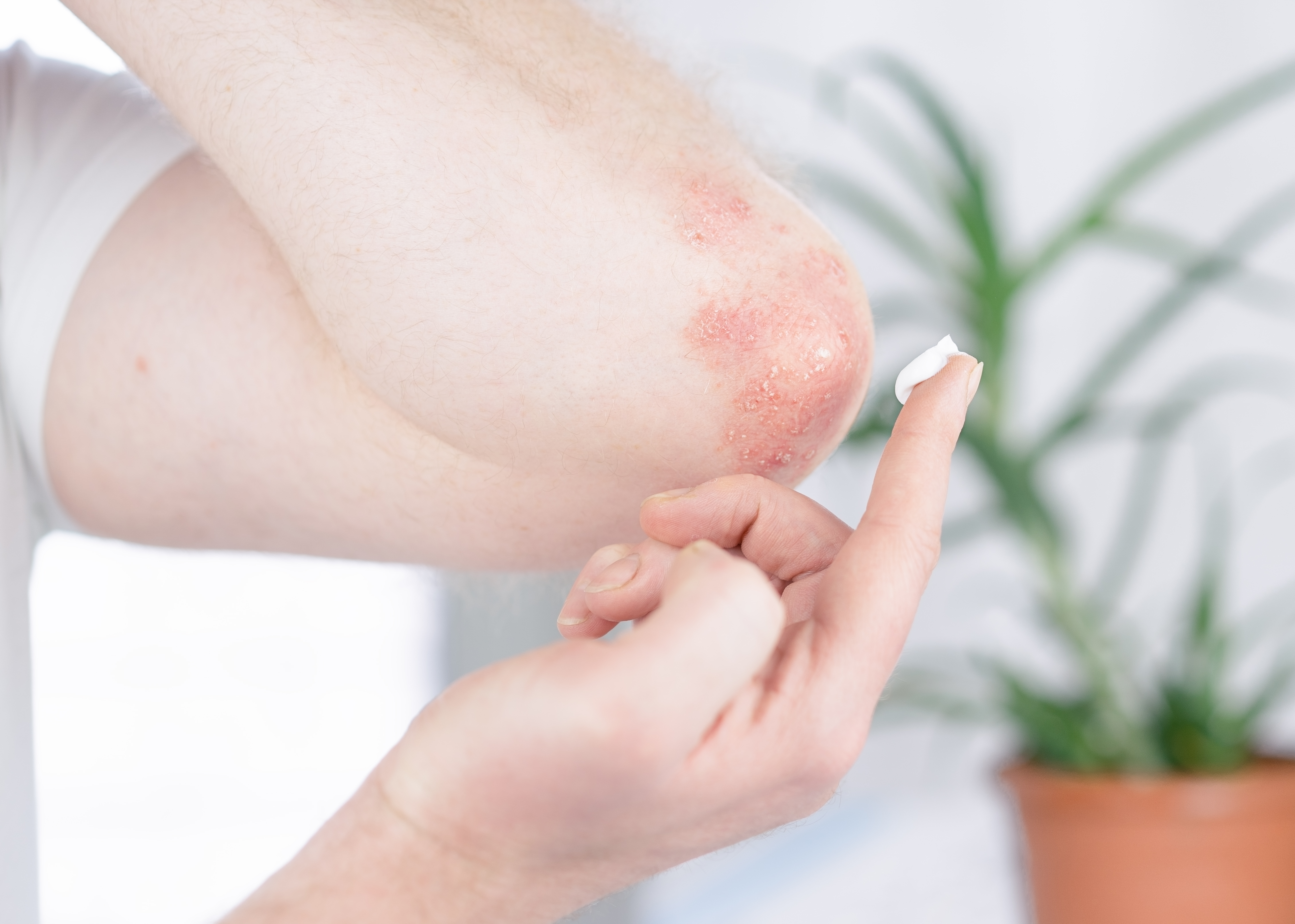
Plaque psoriasis is a chronic, noncontagious skin condition that causes thick, red, dry patches (plaques) on the skin. These plaques can be itchy and covered with silvery scales. Plaque psoriasis most often occurs on the scalp, elbows, and knees on children but can occur on other parts of the body as well. We are recruiting children between the ages of 2 and 17 years old with plaque psoriasis to participate in an investigational study. The purpose of the study is to evaluate the safety and tolerability of topical tapinarof cream, 1% when applied once daily to areas of the skin with plaque psoriasis.
The study consists of applying tapinarof cream, 1% once daily over a 12-week period. This is an open-label study which means that there is no placebo and participants are guarenteed to receive the study treatment. The study consists of a 12-week primary treatment phase and at the end of the study, there is an option for participants to continue use of the treatment for an additional 40-weeks.
For the Primary Treatment Phase, participation will last approximately 17 weeks, including up to 4-weeks for screening, 12 week treatment phase, and a follow-up visit at week 13. . You will have a total of 8 in-person clinic visits, including your initial screening visit and 1 follow-up visit once you have finished receiving the study treatment.
Visits take approximately 1 to 3 hours each, except for the screening, week 4, week 8, and week 12 visits which may take up to 5 hours.
Blood samples will be collected from all participants at each visit.
See if you qualify for the study!





